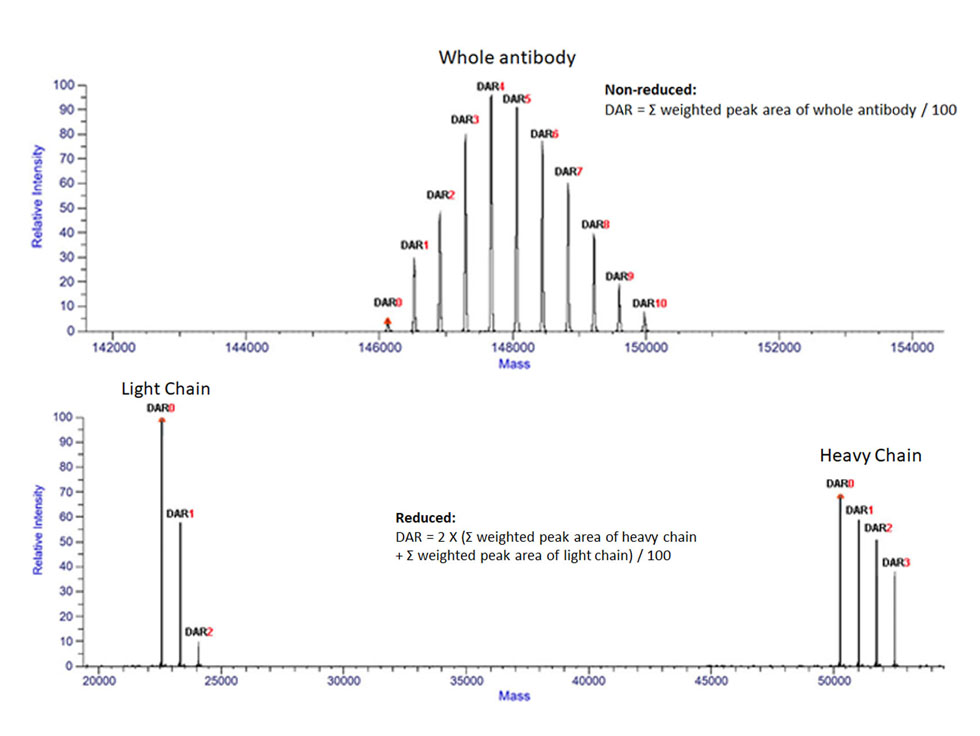
Overview: Drug-antibody ratio (DAR) determination is to measure the average number of drug molecules that are covalently attached to an antibody-drug conjugate (ADC) molecule. The number of drugs is directly related to the efficacy of ADCs, thus the DAR determination is a crucial step for quality control and optimization of synthesis processes in the ADC development. The mass spectrometry is a very powerful and widely used tool to separate different species of ADCs for DAR determination.
Sample preparation: The ADC sample is removed glycans to reduce the complexity. The ADC conjugated by disulfide bonds is usually treated by Dithiothreitol to separate light chain and heavy chain.
High-performance liquid chromatography – mass spectrometry (HPLC-MS) analysis: The ADC sample is desalted and separated using reversed phase (RP) or SEC (Size exclusion chromatography) high-performance liquid chromatography (HPLC). The ionized ADC molecules are then analyzed using a mass spectrometer to determine the mass-to-charge ratio (m/z).
DAR calculation: First, the raw mass spectra data is deconvoluted to obtain the true mass of each molecule in the sample. The DAR is then calculated by analyzing the intensity of each ADC species.